 There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. Please note that the elements do not show their natural relation towards each other as in the Periodic system. A higher atomic weight than the one on its left. Mendeleev found he could arrange the 65 elements then known in a grid or table so that each element had: 1. The first chemical element is Hydrogen and the last is Ununoctium. The periodic table we use today is based on the one devised and published by Dmitri Mendeleev in 1869. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. This list contains the 118 elements of chemistry. Plant Inspection & Process OptimalisationĬhemical elements listed by atomic number The elemenents of the periodic table sorted by atomic numberĬlick on any element's name for further chemical properties, environmental data or health effects.Therefore, there are various non-equivalent definitions of atomic radius. There is also a 2019 edition of this table. #ATOMIC TABLE PDF#Here is the pdf file of the color periodic table so you can save and print it. Every element has a unique combination of period and group, and a unique atomic number. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. Periods are the rows of the periodic table, groups are the columns. 
#ATOMIC TABLE FREE#However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. 2013 Edition This free periodic table wallpaper has a white background. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. The Hampton features a modern contemporary Grey Wood grain finish with heavy-duty quality construction. The Hampton 3-in-1 combination table includes billiards, table tennis, and a Dining table with dual storage bench seating all in one. It must be noted, atoms lack a well-defined outer boundary. Add the ultimate multi-functional table to your game room that is ideal for the entire family. The atomic radius of Germanium atom is 122pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Germanium are 70 72 73 74. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. You’ll also notice that each element’s atomic mass increases as you move across the table. The atomic number is how many protons the element’s atom possesses. The elements are ordered by their atomic numbers, which increase as you move across and down the periodic table. 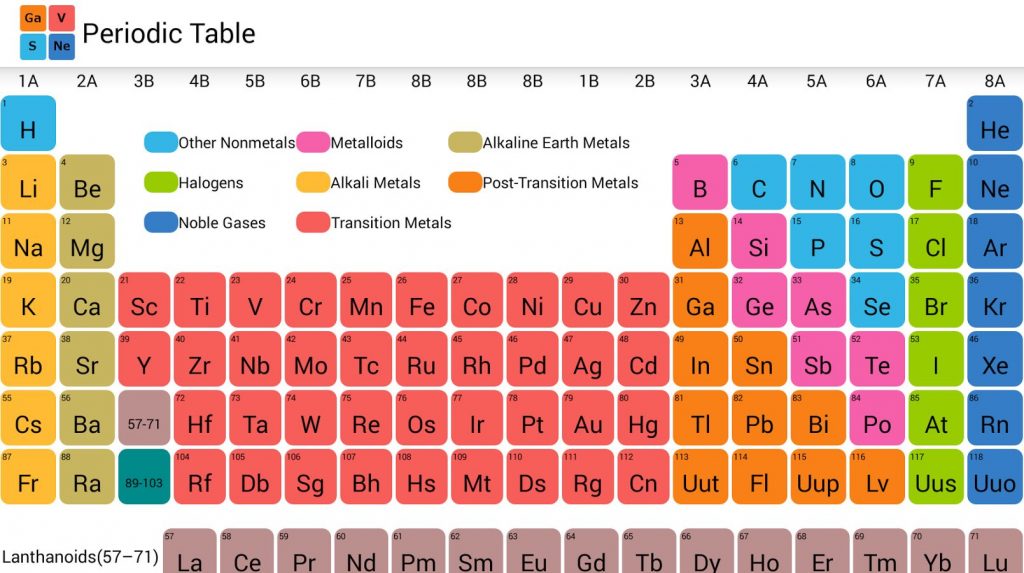
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Read the periodic table from top left to bottom right. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol Z. The modern day periodic table has elements arranged according to. In the periodic table, the elements are listed in order of increasing atomic number Z. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Periodic table of the chemical elements The periodic table, also known as the periodic table of the ( chemical) elements, is a tabular display of the chemical elements. We now know that chemical properties are due to electronic configuration and not atomic mass. Germanium is a chemical element with atomic number 32 which means there are 32 protons in its nucleus. Atomic Number – Protons, Electrons and Neutrons in Germanium 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
